I am not a doctor, scientist, or expert in anything. This content should not be construed as advice or recommendation, but is intended for entertainment and informational purposes only.
Watching football the other day I saw an ad for Pfizer’s new RSV “vaccine” that’s been cooked up and is ready to jab into waiting arms. After reading through the package insert on this new product, one can see that Pfizer is following much of the same playbook used in the development of the COVID shots.
It’s also apparent that the definition of vaccine has been changed for good so as to no longer mean what we thought it meant for most of vaccine history. Previously, we were led to believe that if we got a vaccination we were protected from the pathogen and the resulting disease, and we couldn’t transmit the pathogen thereby protecting others. New vaccinations appear to only need to possibly reduce severe disease, not prevent infection or transmission.
According to the CDC, RSV (respiratory syncytial virus) is a common respiratory virus that typically causes mild, cold-like symptoms that most people recover from within a week or two although RSV can become more severe in infants and the elderly. Pfizer’s new shot, ABRYSVO, is indicated for pregnant women and adults over 60 years of age.
The reason for giving this shot to pregnant women is not to protect them, but to protect the unborn fetus from birth to 6 months of age against lower respiratory tract disease (LRTD) and severe LRTD. I have to give Pfizer credit, they’ve found a way to get another vaccination into children without them even needing to be born.
So how was the shot tested and does it even perform as advertised?
I’ll get straight to the punchline to spare you all of the reading, but I would encourage yo to read the entire article to get a better handle on the scope of fuckery involved.
First, the two phase 3 clinical trials in the package insert, which are still ongoing, did not use saline placebos in their placebo groups. Instead, the shot without the antigen was used as placebo. Is the shot safe? Who knows and who cares?
Second, the shots were not tested to determine if they prevent infection or transmission.
Third, while Pfizer claims to have met most of the endpoints for demonstrating vaccine efficacy, to say the results are underwhelming would be a massive understatement.
Assuming Pfizer’s data is legitimate, a massive leap of faith given that we will never see the raw data, roughly 80 pregnant women would need to get these shots to prevent 1 case of severe LRTD for infants 6 months or younger, and you would need to vaccinate over 140 pregnant women to prevent 1 hospitalization among infants 0 to 6 months of age.
You would need to vaccinate more than 760 people over the age of 60 to prevent 1 case of LRTD with 2 or more symptoms, and over 1,300 to prevent 1 case of LRTD with 3 or more symptoms. Vaccination does virtually nothing when it comes to protecting against severe LRTD in this age group.
Efficacy, much like beauty I suppose, is in the eye of the beholder. Read on if you want more of the story.
Referring back to the package insert, four clinical studies were performed, two of which are still ongoing. How Pfizer can claim anything while studies are ongoing is beyond me, but we’ve learned that shots in arms trump waiting for the data which will be manipulated and ignored anyway.
The first study is an ongoing Phase 3, randomized, double-blind, multi-center, placebo-controlled study being performed on roughly 7,357 pregnant women with uncomplicated, singelton (one fetus) pregnancies. All the fancy words must mean this is a great study so we shouldn't feel the need to read any further. But we will.
PLACEBO CONTROLLED?
What is the placebo being used for the 3,675 women in the placebo group? True to form, Pfizer’s version of placebo is not the inert saline placebo but is instead:
0.5 mL dose, containing the same buffer ingredients in the same quantities as in a single dose of ABRYSVO.
What are these buffer ingredients?
ABRYSVO also contains the following buffer ingredients: 0.11 mg tromethamine, 1.04 mg tromethamine hydrochloride, 11.3 mg sucrose, 22.5 mg mannitol, 0.08 mg polysorbate 80, and 1.1 mg sodium chloride per 0.5 mL. ABRYSVO is a sterile, clear, and colorless solution
Tromethamine is a drug used to prevent or treat acid problems in the blood, and is administered intravenously as an infusion over time. Side effects include:
Signs of an allergic reaction, like rash; hives; itching; red, swollen, blistered, or peeling skin with or without fever; wheezing; tightness in the chest or throat; trouble breathing, swallowing, or talking; unusual hoarseness; or swelling of the mouth, face, lips, tongue, or throat.
Signs of low blood sugar like dizziness, headache, feeling sleepy, feeling weak, shaking, a fast heartbeat, confusion, hunger, or sweating.
Signs of high potassium levels like a heartbeat that does not feel normal; feeling confused; feeling weak, lightheaded, or dizzy; feeling like passing out; numbness or tingling; or shortness of breath.
Trouble breathing, slow breathing, or shallow breathing.
Shortness of breath, a big weight gain, or swelling in the arms or legs.
Fever.
This medicine may cause tissue damage if the drug leaks from the vein. Tell your nurse if you have any redness, burning, pain, swelling, blisters, skin sores, or leaking of fluid where the drug is going into your body.
Notice the last bullet point. If tromethamine leaks from the vein it can cause tissue damage yet this is going to be injected intramuscularly. Hmmm, wonder why? Ah yes, so it can stimulate an immune response. Does this match your definition of a placebo?
So we can immediately answer the question of whether or not this shot is safe for women and unborn children. They have no fucking idea if it’s safe which is the point of these studies.
Once it’s approved and on the market you can’t go back to test against a real placebo because it would be unethical to give someone a placebo when there is already an approved intervention available regardless of the fact that it was never tested against a placebo for safety. Get it?
The package insert informs us that there were more pre-term births in the ABRYSVO group vs placebo (5.7% to 4.7%), more adverse events in infants from birth to 1 year of age (37.1% compared to 34.5% in placebo), low birth rate in 5.1% of ABRYSVO group vs 4.4% of placebo group, and jaundice in 7.2% of the ABRYSVO group vs 6.7% in placebo group.
Maternal adverse events were also higher in the ABRYSVO group and there were more fetal deaths among the ABRYSVO group although they claim higher overall mortality (fetal, neonatal, post-neonatal) and congenital abnormalities for the “placebo” group. Unfortunately we don’t have the raw data and no one will likely ever see it, so we can’t confirm any of this data beyond what has been provided.
Since the placebo was effectively the same formula as the vaccine sans antigen, nobody can say whether or not this shot is safe for women and their unborn children, but that’s not nearly as important as getting the shot into people. Revenues to be made and shareholders to appease you know?
What about the other studies?
The second study was a Phase 2, randomized, placebo-controlled, observer-blind study investigating the safety of two doses levels (120 mcg and a higher dose) of ABRYSVO administered to pregnant women. The package insert only mentions the 115 women receiving the 120 mcg dose. Little is mentioned about this trial but you can read more about it at https://clinicaltrials.gov/study/NCT04032093?a=14.
This study actually had four treatment arms. One group (115 participants) received a 120 mcg doses, the second (117 participants) received 120 mcg dose with Aluminum Hydroxide, the third group (116) received a 240 mcg dose, and the fourth group (114) received 240 mcg with Aluminum Hydroxide. The placebo group had 117 participants.
Interestingly, the placebo group had more people not complete the study and also had worse outcomes in regard to serious adverse events and obstetric complications for pregnant women compared to the 120 mcg group. Infants born to women in the placebo group had the same or worse outcomes compared to the treatment group, except for serious adverse events, within 12 months of age.
Are we supposed to believe that 120 mcg of ABRYSVO is protective against adverse events and obstetric complications? I smell a rat in this data.
There was an additional phase 2 “placebo-controlled" study performed on expectant mothers that involved injecting them with various dosages of the RSV vaccine plus Tdap vaccine concomitantly. The “placebo” group received an unspecified placebo plus the Tdap vaccine. Placebo-controlled? Nice try Pfizer.
OVER 60
The third study referenced in the package insert is an ongoing, multi-center, randomized, placebo-controlled study to assess the efficacy and safety of ABRSYVO in people over 60 years of age. 17,215 received ABRYSVO and 17,069 received the same placebo that is not a placebo that the pregnant women in study 1 received.
Adverse events events between the two groups were similar, but what would one expect when you are essentially receiving the same shot.
It would be worth mentioning that ABRYSVO has not been evaluated for the potential to cause carcinogenicity, genotoxicity, or impairment of male fertility. But the upside is that a developmental toxicity study in female rabbits revealed no evidence of impaired female fertility after administration of a vaccine formulation containing two times the antigen content of a single human dose of ABRYSVO. So I guess there’s that.
DOES IT WORK?
Silly, you’re asking the wrong question. You should be asking if these shots result in antibodies being produced which it appears they do.
But since you asked, let’s answer the question.
Vaccine efficacy (VE) was defined in study 1 as:
the relative risk reduction of the endpoints of severe LRTD caused by RSV and LRTD cause by RSV in infants born to individuals who received ABRYSVO compared to infants born to individuals who received placebo.
So, does it work? Depends on what you mean by “work.” According to the results:
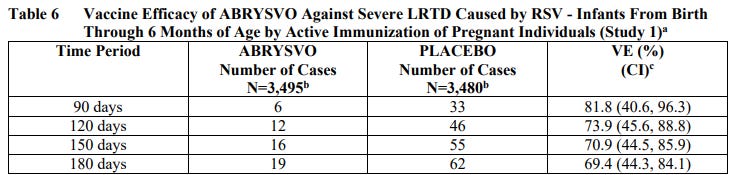
The VE results met the statistical criterion for success (a CI lower bound >20%) for reducing severe LRTD due to RSV, at all timepoints to within 180 days. The VE results did not meet the statistical criterion for success (a CI lower bound >20%) for reducing LRTD due to RSV; however, clinically meaningful efficacy was observed after 90 days through 180 days after birth.
Absolute risk reduction (ARR) of severe LRTD in the ABRYSVO group was 1.23% (62/3480 - 19/3495) at 180 days after birth.
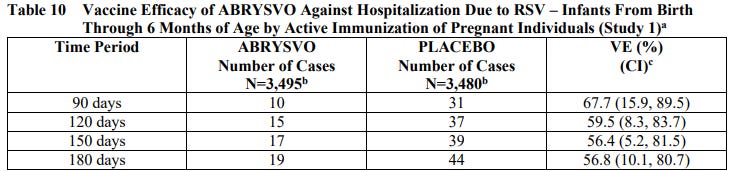
ARR of hospitalization in the ABRYSVO group was 0.71% (44/3480 - 19/3495) at 180 days after birth.
Assuming this data is legitimate, roughly 80 pregnant women would need to get these shots to prevent 1 case of severe LRTD, and you would need to vaccinate about 140 pregnant women to prevent 1 hospitalization among infants less than 6 months of age.
For those over the age of 60, vaccine efficacy was defined in study 3 as:
the relative risk reduction of first episode of RSV-LRTD in the ABRYSVO group compared to the placebo group in the first RSV season.
The study met the pre-specified success criteria for demonstration of efficacy of ABRYSVO for the primary objectives of prevention of RSV-LRTD with ≥2 symptoms and prevention of RSV-LRTD with ≥3 symptoms
According to Pfizer, this product is also successful for people over the age of 60. How good does it work?
The ARR for LRTD with 2 symptoms was 0.13% (33/16308 - 11/16306) and was 0.073% (14/16,308 - 2 /16,306) for LRTD with 3 symptoms. Plus there were 2 cases of RSV with severe LRTD in the placebo group vs none in the ABRYSVO group which is basically an ARR of 0.0% for reduction of severe LRTD.
LRTD from RSV among this age group doesn’t appear to be particularly prevalent, and it’s mostly a mild disease since there were only two cases among the 32,000+ participants who experienced severe LRTD.
Again, assuming this data is legitimate, you would need to vaccinate more than 760 people over the age of 60 to prevent 1 case of LRTD with 2 or more symptoms, and over 1,300 would need the shots to prevent 1 case of LRTD with 3 or more symptoms. Vaccination effectively does nothing when it comes to protecting against severe LRTD, which is virtually non-existent, in this age group.
SHADES OF COVID
Notice that neither of the efficacy end points for these two studies had anything to do with preventing infection or transmission. The COVID plandemic laid bare the the lie about many of the vaccines people receive under the illusion they protect the recipient from infection and eliminate his or her ability to transmit a virus to others.
In true Orwellian fashion, the fact that flu, COVID, RSV, and other vaccines don’t prevent infection or transmission, and at best may reduce the severity of disease slightly has not caused pause among the Big Pharma/Public Health apparatus which is doubling and tripling down on these failed interventions. When the lies are finally made evident, they just rewrite history.
Now, we are supposed to believe that it was common knowledge all along that vaccines only reduce severe disease somewhat, and not infection or transmission. If you sometimes wonder if you’re the one going crazy, you’re not. This is gaslighting at its finest.
It should be someone’s choice as to whether they want to vaccinate themselves or their children (or unborn children), but they should be presented with the true risks and benefits in order to make their own informed choices. Unfortunately, you can’t trust “The Science.”